Compensatory degradation of cytosolic misfolded proteinsmediated by the interaction
between mitophagy receptor FUNDC1 and cytosolic chaperone HSC70
Supported by the Major Project of the National Natural Science Foundation of China (31790404) and other awards, Quan Chen, a professor at Institute of zoology, Chinese Academy of Sciences, and his fellows reported a new protein degradation pathway mediated by mitochondrial protein FUNDC1 and cytosolic chaperone HSC70. In this pathway, mitochondria help cells to maintain the overall proteostasis by employing their own protease or mitophagy machinery. Their research was published online in the EMBO journal on December 27th 2018 entitled “A Mitochondrial FUNDC1/HSC70 Interaction Organizes the Proteostatic Stress Response at the Risk of Cell Morbidity”. Link: http://emboj.embopress.org/content/early/2018/12/21/embj.201798786
Both protein quality and mitochondrial quality are vital to basic cell viability. Protein homeostasis, or proteostasis, is maintained through a coordinated network including chaperones and two proteolytic systems, the ubiquitin-proteasome system and the autophagy-lysosome system. As multitasking cellular organelles that are essential for diverse functions including energy production and cell metabolism, mitochondria have relatively independent protein quality control systems, including free radical scavengers, chaperones, AAA proteases, fusion and fission machineries and mitophagy machineries. Impaired proteostasis and mitochondrial dysfunction are both causally linked with aging and age-related disorders. They may reinforce each other, but the mechanism of their correlation is unclear.
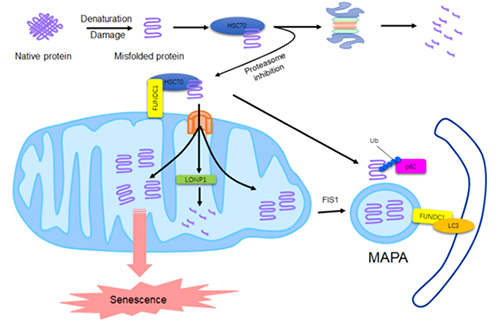
Quan Chen’ group found that the outer mitochondrial membrane protein FUNDC1, a previously reported mitophagy receptor, interacts with the chaperone protein HSC70 to mediate the mitochondrial translocation of cytosolic misfolded proteins. These proteins are then imported via the TOM-TIM complex into the mitochondrial matrix where they can be degraded by LONP1 protease. In proteasome inhibited cells, the interaction between FUNDC1 and HSC70 is enhanced, and the unfolded proteins recruited to the mitochondria are increased accordingly. The proteins accumulated in the mitochondrial matrix will concentrate in certain region and promote the formation of a specific Mitochondrion-Associated Protein Aggregates (MAPAs). This FUNDC1/HSC70-pathway help cells to maintain proteostasis by employing the mitochondrial protease or mitophagy machinery, but excessive accumulation of unfolded proteins in mitochondria result in mitochondrial disintegrity, AMPK activation and cellular senescence. We suggest that mitochondrial organize the compensatory degradation of cytosolic misfolded proteins at the risk of their own malfunction and cell morbidity.
This research uncovers a new link between impaired proteostasis, mitochondrial dysfunction and cellular senescence. It also provides a new explanation for aging and age-related diseases, and a new theoretical guidance for the treatment and prevention of these diseases.

Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@donnasnhdiary.org
京ICP备05002826号 文保网安备1101080035号 Copyright 2017 NSFC, All Right Reserved